Most of us know someone who has taken antidepressants. But psychedelic drugs? Not so much. Many people believe they are illegitimate and dangerous. You might be surprised to hear that psychedelic drugs like MDMA and LSD have a lot in common with antidepressants. They both work with the same neurotransmitter in the brain: serotonin.
And indeed, antidepressants and psychedelic drugs promise to heal similar mental illnesses and can also have similar side effects. Do you know how they work in the brain? No? Good! That’s exactly what this article is about. Before we can talk about your brain on these drugs, though, it’s important to have a basic understanding of the brain and its serotonergic system. Don’t worry, it’s super fascinating stuff, and easy as 1-2-3:
1. Brain basics
Below, we’ll talk about neurotransmitters, synapses and chemical signaling. If these things are even vaguely familiar to you, then read on. If not, I recommend reading part 2 of Tim Urban’s fantastic—and highly entertaining—Neuralink and the Brain’s Magical Future story on the Wait But Why blog. You’ll learn everything from brain anatomy to neural networks in just 15 minutes.
How neurons communicate
When a neuron fires, the cell body (soma) sends an electrical signal down its axon to its axon terminals. This is where one neuron connects to the dendrites of the next neuron. In between is the synapse. From the axon terminals, a chemical signal activates the dendrites and sends a message to the soma of the next neuron. The soma collects the messages and once a threshold is exceeded, it fires off an electrical signal down its own axon and the process repeats.
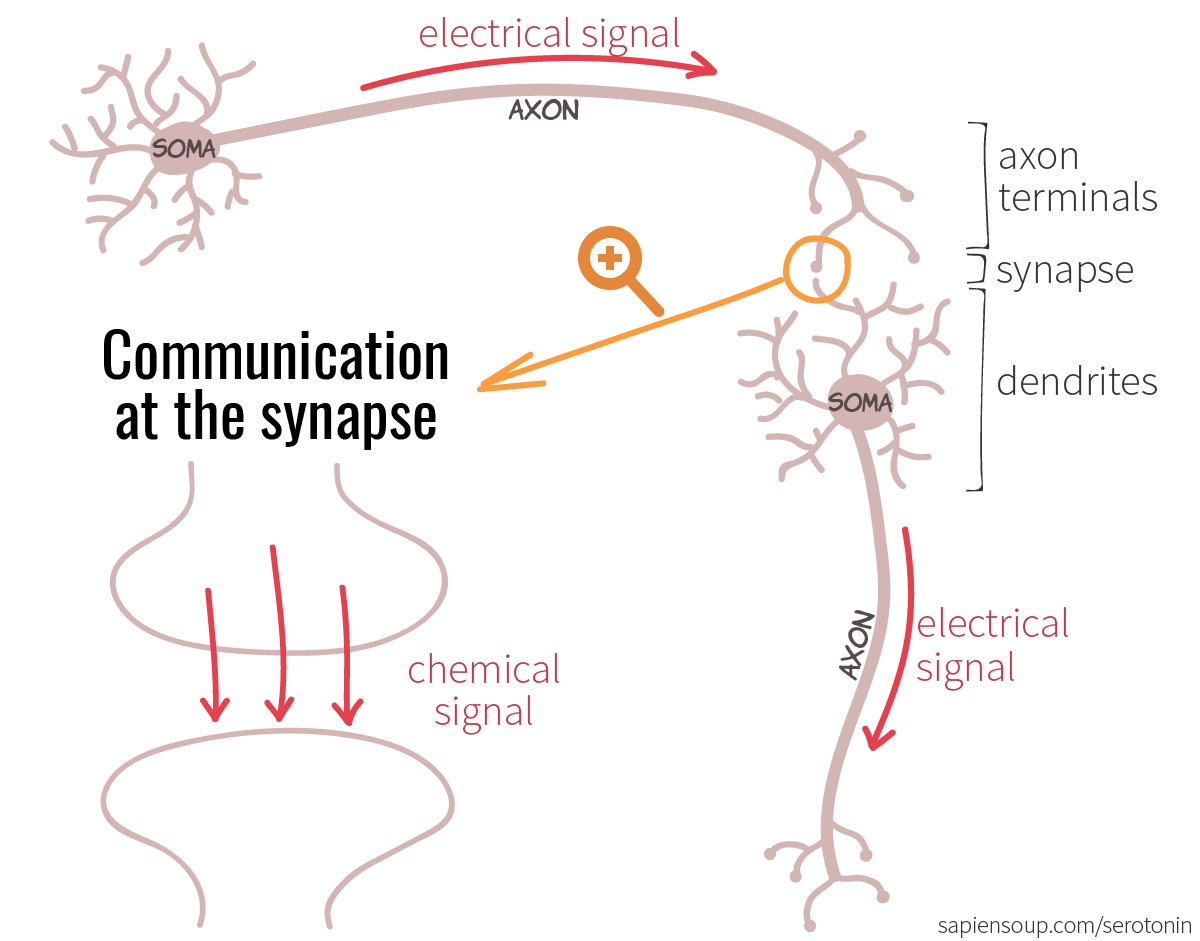
Chemical signals are made from neurotransmitters. How they are produced, sent and received is the key to understanding the interactions between drugs and the serotonergic system.
Neurotransmitters
You have probably heard of the neurotransmitters dopamine and serotonin. A simplistic view would be that dopamine regulates feelings of reward and serotonin regulates feelings of happiness. Yes, it’s more complex than this, but we’re just getting started.
We have more than 100 different types of neurotransmitters in our brain and their job is facilitating the communication between neurons. Think of a neurotransmitter as a language: some neurons speak dopamine, others speak serotonin and so on. While some neurons are multilingual, let’s say fluent in serotonin and dopamine, most of them speak just one language. All neurons which speak serotonin make up the serotonergic system.
2. The serotonergic system
The serotonergic system is amongst the oldest neurotransmitter systems in the brain. It might be as old as 750 million years; even single-celled organisms carry serotonin receptors.1 2 In humans, those neurons originate from the raphe nuclei in the brainstem and form a network spanning every corner of the brain and influencing nearly every aspect of our lives. It plays a key role in regulating mood, sexual behavior, aggression, impulsivity, cognitive function, appetite, pain, thermoregulation, circadian rhythm, sleep and memory. 3 4

All those substances do essentially one thing: they raise the serotonergic activity in the brain. Why? Because raising serotonergic activity makes you happy, social and active; whereas lowering serotonergic activity makes you depressed, irritable and more prone to mental illnesses.
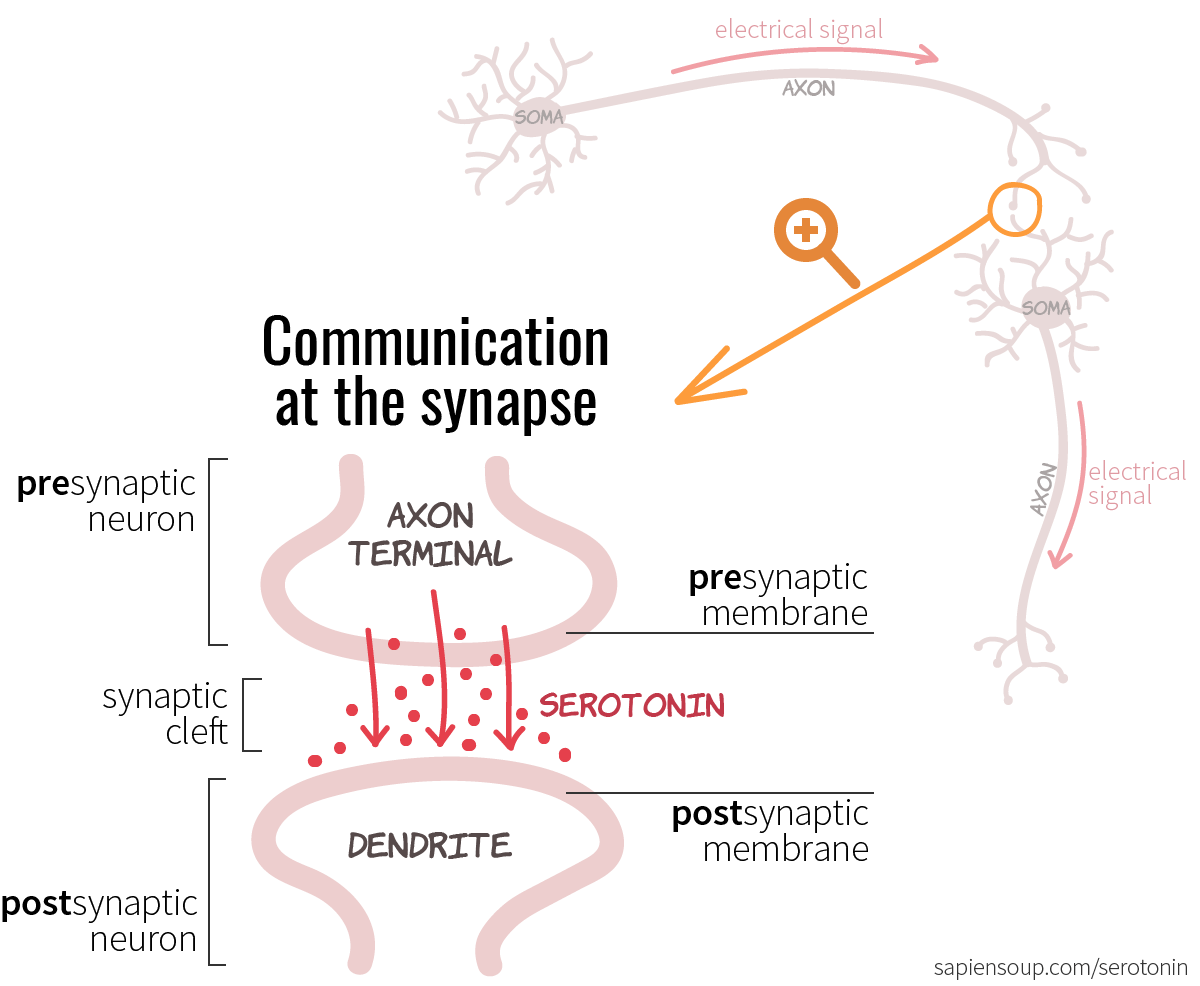
Serotonin in the synapse
This is where it gets really interesting. Before we dive into the life of a serotonin molecule, let’s make sure we’re all on the same page. Take another look at the more detailed version of how communication happens at the synapse. On the top is the axon terminal of the “sender-neuron” which is often referred to as the presynaptic neuron. On the bottom is the “receiver-neuron”—the postsynaptic neuron. The skin of the neurons is the membrane; and the little gap in between is called the synaptic cleft. What gets sent from the sender to the receiver? A chemical signal, otherwise known as neurotransmitter; and in the case of a serotonergic neuron the neurotransmitter is serotonin.
Now we get to the nitty gritty. The following graphic illustrates the lifecycle of a serotonin molecule. Follow the orange dots from one to seven and check out the explanation below.
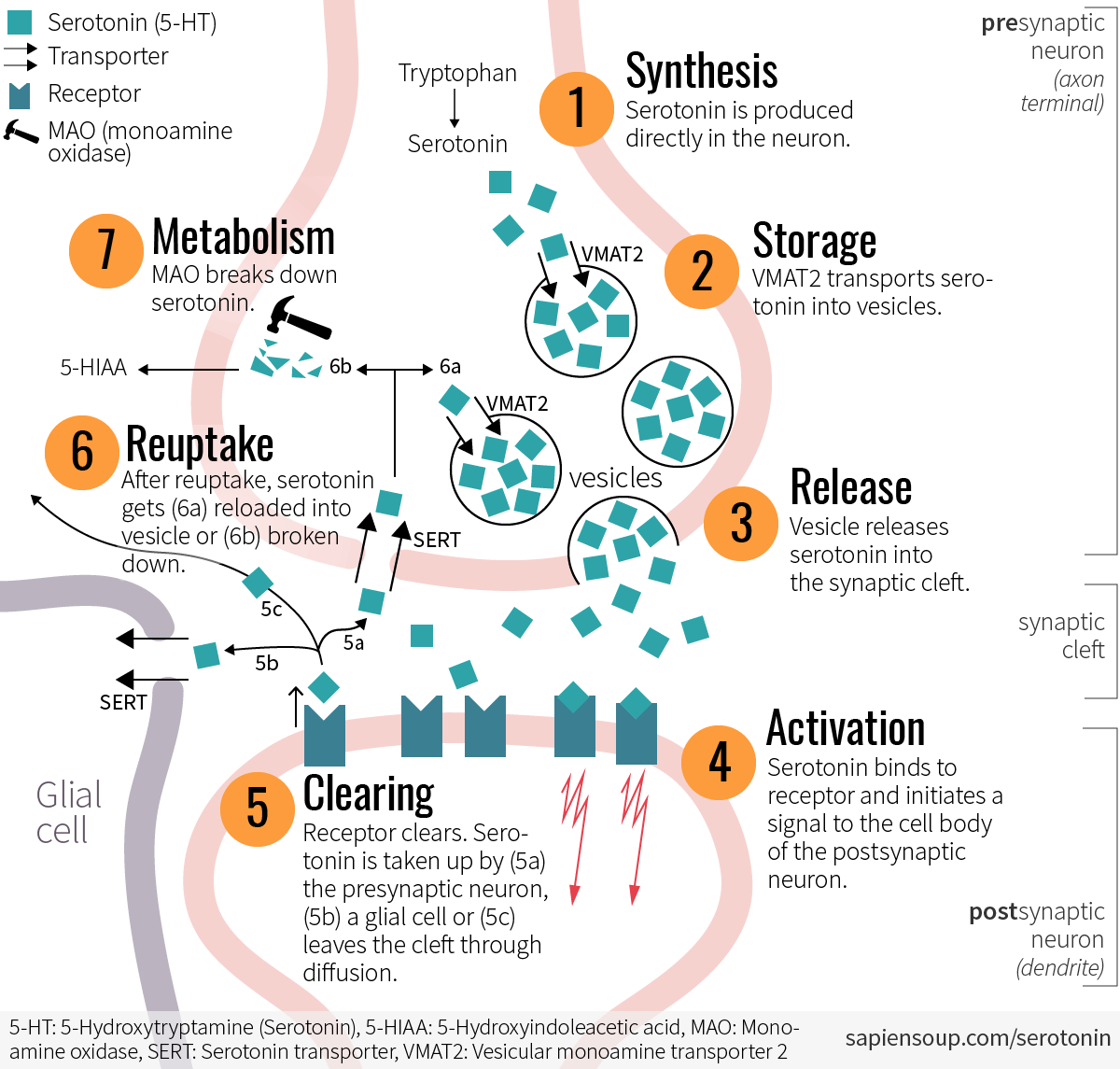 Signaling in chemical synapses
Signaling in chemical synapses
1 Synthesis
Well over 90 percent of the serotonin in our body is made in our gut. But since serotonin can’t cross the blood-brain barrier, it has to be synthesized in the brain from scratch. What does cross the blood-brain barrier however is tryptophan, the fundamental building block of serotonin.6 Within the neuron, enzymes turn tryptophan into 5-HT which is the chemical name for serotonin.7
How does our body get tryptophan in the first place? Tryptophan is contained in certain foods, particularly proteins. You may have heard that turkey is rich in tryptophan—so is every other kind of meat, as well as cheese, dairy products and eggs. Paradoxically, eating a protein rich diet is not necessary useful for a steady tryptophan supply in the brain. Why? Read my story about amino acid competition at the blood-brain barrier.
2 Storage
Serotonin is stored in tiny bubbles—only 50 nanometers in diameter—called vesicles. How does it get in there? Initially, the serotonin floats in the cytosol, the fluid within the neuron. A transport protein called VMAT2 fishes the serotonin out of the cytosol and channels it into one of the vesicles. The vesicles then travel closer towards the synaptic cleft and wait for their signal.
3 Release
When signalled, the vesicles meld with the cell membrane in a process called exocytosis. The serotonin gets released into the synaptic cleft.
4 Receptor activation
When serotonin binds to the receptors of the postsynaptic neuron, each receptor sends off a signal to the cell body of the neuron. When enough of these signals accumulate, the postsynaptic neuron fires, causing an electrical signal to travel down its axon to its own axon terminals, in turn causing a release of serotonin that stimulates the next neuron. This chain reaction cascades through any number of neurons….
5 Receptor clearing
Where does a serotonin molecule go after it has activated a receptor? There are a few options: (a) it may get taken back up into the presynaptic neuron; (b) it may get taken up by a neighboring glial cell (glial cells are the most abundant cells in the brain—they don’t transmit signals but they do help keep everything neat and tidy); or (c) it may get diffused away from the synaptic cleft via extracellular fluid.6
6 Reuptake
Along the presynaptic membrane are serotonin transporters (SERT) that pull serotonin back into the cell in a process called reuptake. These transporters are basically groups of proteins that act like a gate: one in—one out. One molecule binds to the transporter on the outside of the membrane and changes the transporter’s configuration. Consequently, another molecule drops off, but on the inside of the membrane.8

Back in the presynaptic neuron, some of the serotonin gets reloaded into vesicles and will be reused. Producing serotonin from scratch is a complex process and takes time. Therefore, recycling helps the brain maintain a steady supply.
7 Metabolism
Any remaining serotonin gets broken down by the enzyme MAO (monoamine oxidase) and excreted from the cell as the metabolite 5-HIAA (5-Hydroxyindoleacetic acid).
How serotonin neurons avoid overstimulation
The brain can’t produce large quantities of serotonin at once, therefore it doesn’t release large quantities of serotonin at once either. In fact, serotonergic neurons have multiple ways of up- and downregulating their serotonin response in order to maintain balance and protect themselves from overstimulation.
Follow the orange dots below to see a few examples of these protection strategies.
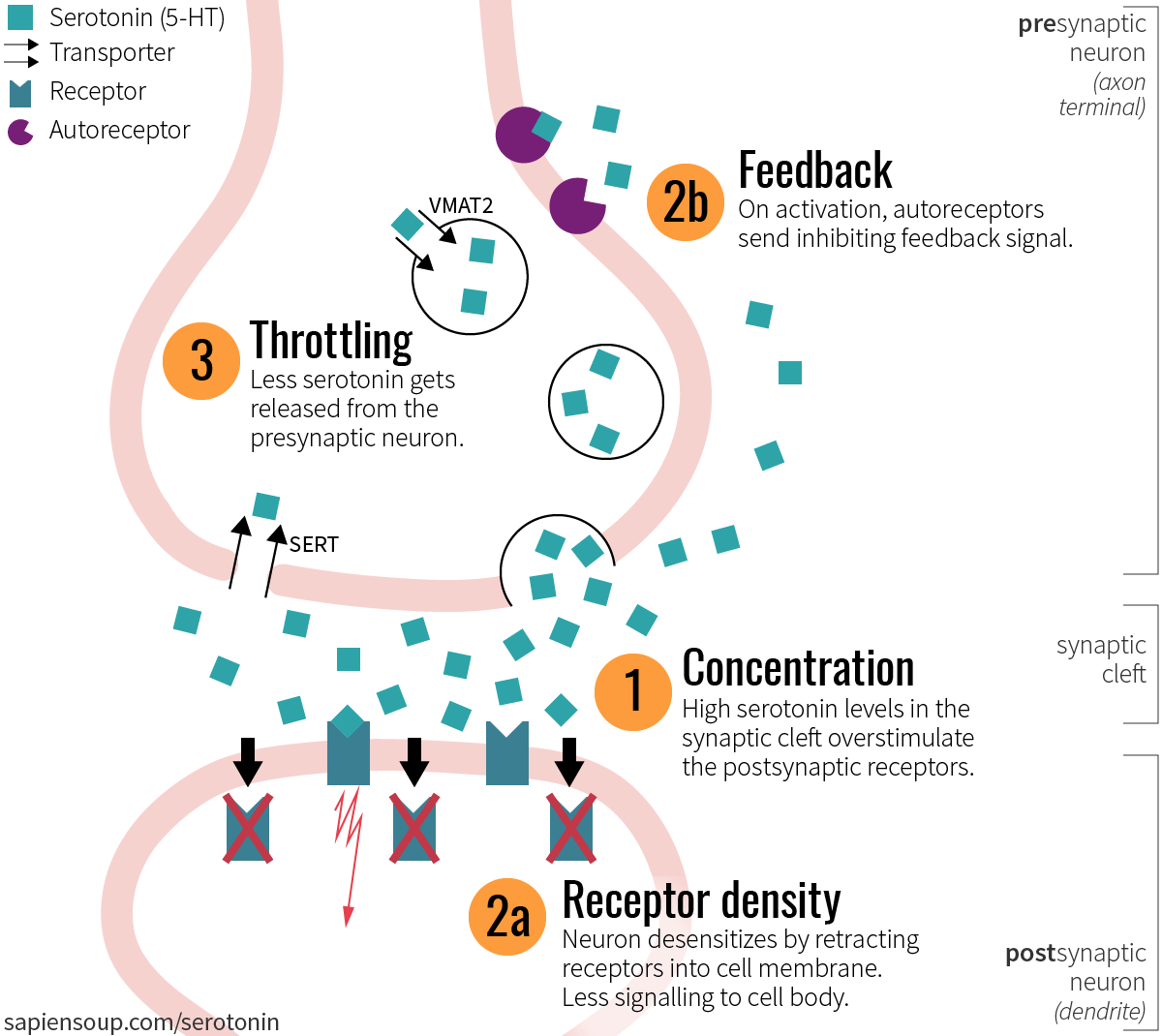
If there is (1) a high concentration of serotonin outside the neuron, the neuron reacts with
(2a) Reducing receptor density
Amazingly, a neuron can cause its receptors to retract behind the synaptic membrane, putting them out of reach of being activated by over-abundant serotonin. With fewer receptors available, fewer activations occur, and the neuron is in turn less likely to fire off a signal.9
(2b) Feedback via autoreceptors
Receptors are not only found on the postsynaptic membrane. Some are located on the axon terminals or even directly on the soma of a neuron. If too much serotonin is floating around in the brain and these autoreceptors get activated, they send an inhibitory signal to the presynaptic neuron that causes it to (3) throttle the release of serotonin.6
The versatility of serotonin receptors
Do you recall from the beginning how serotonin regulates mood, sexual behavior, cognitive function, sleep, memory and so on? How does it accomplish all that? Well, in reality there isn’t just one single type of serotonin receptor—there are 14. They are numbered from 1 to 7 and further categorized into A, B, C, etc. Remember, the chemical name for serotonin is 5-HT. Going forward we’ll talk a lot about 5-HT2A receptors, since they are the target of hallucinogenic drugs like LSD and psilocybin.
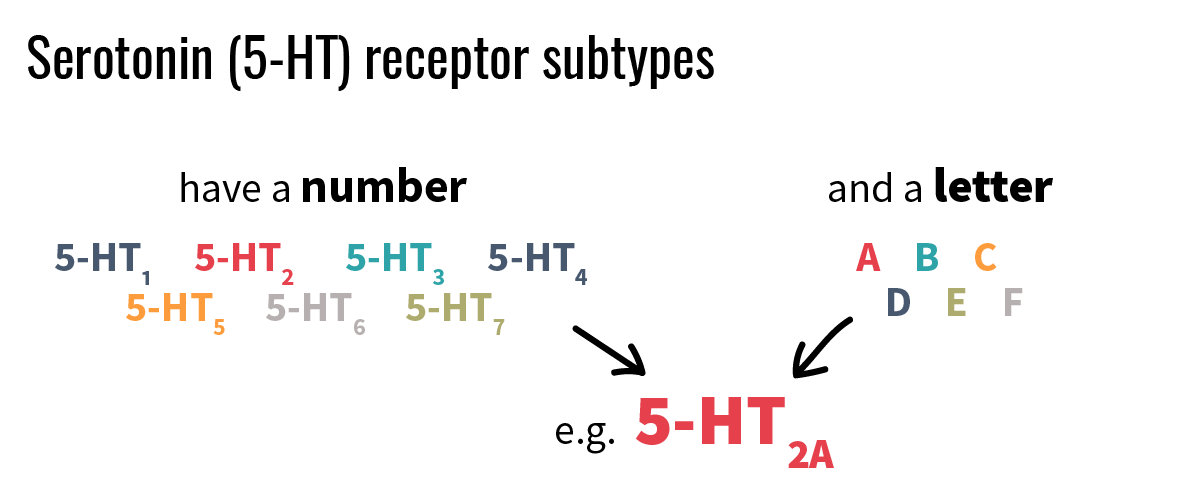
All 5-HT subtypes possess special qualities in how they regulate mood, anxiety, impulsivity, aggression, migraines, etc. Some of these subtypes act as regular receptors at the postsynaptic membrane, while others act as autoreceptors on the axon terminals, dendrites or directly on the cell body. The 5-HT2A receptor, for example, is a receptor on the postsynaptic membrane and regulates mood, anxiety and schizophrenia.10 Wikipedia offers a fantastic overview of 5-HT receptor subtypes if you wish to go deeper.
3. How antidepressants & psychedelic drugs stimulate the serotonergic system
Now that you know how serotonin acts in the synaptic cleft it will be easy for you to understand the mechanisms of antidepressants like SSRIs and MAOIs as well as psychedelic drugs like LSD, psilocybin and MDMA. Each of these substances stimulate serotonergic neurons, but each in different ways.
Antidepressants
Before 1950 it was believed that mental illnesses like schizophrenia or autism were caused by “refrigerator mothers”—mothers who were emotionally distanced and cold with their offspring.11 The psychiatric community had no idea that behaviour patterns, such as schizophrenic or autistic behaviour, might arise from neurochemical events in the brain.1
In the late 1930s, serotonin was first discovered in the gut where it played a role in muscle contraction. It took another 15 years before it was detected in the brain—which was in 1953—but still only in the context of muscle contraction. One year later, in 1954 two scientists noticed the chemical similarity between serotonin and LSD.
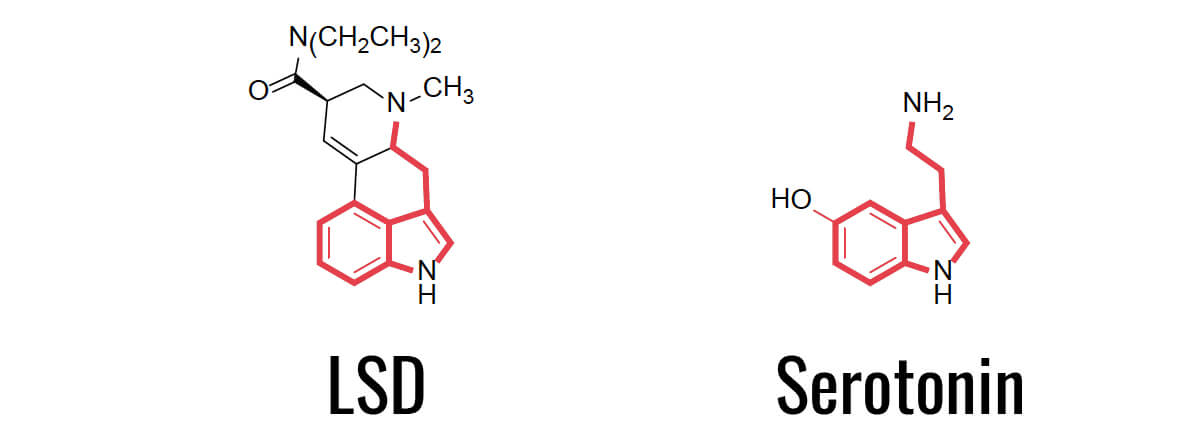 Chemical structure of LSD and serotonin2
Chemical structure of LSD and serotonin2
They had already known that LSD had peculiar effects on mind and behavior, because Sandoz Laboratories had marketed LSD as a psychiatric drug since 1947. Putting one and one together, these two scientists suggested that serotonin might play an important role in mental illness.12
“If neuroscience can be said to have a beginning, one could argue that it occurred in 1954, with the idea that the action of LSD might be related to its effects on the brain serotonin system.”2
After it became obvious, that serotonin was deeply involved in mental sanity it quickly became the center of attention of pharmaceutical companies. Understanding the mechanism by which mood is regulated allowed pharmacologists to experiment with ways to influence it. One result has been the creation of many antidepressant drugs. Here is how they work.
SSRIs: Blocking serotonin reuptake
Selective serotonin reuptake inhibitors (SSRIs) are the most commonly prescribed antidepressant drugs today. You’ve probably heard of Prozac, Celexa, Lexapro, Seroxat or Zoloft. They are all SSRIs.
SSRIs bind to serotonin transporters (SERT) on the presynaptic membrane and block them. This means serotonin can’t get taken back up into the presynaptic neuron. More serotonin remains in the synaptic cleft where it continues to bind to receptors and activates them.

MAOIs: Blocking metabolism
Monoamine Oxidase Inhibitors (MAOI) are older antidepressants, which are still in use, but not commonly prescribed because of their potentially lethal effects. MAOIs keep serotonin from being metabolized and excreted from the neuron, which in turn increases its availability.
Psychedelic drugs
Antidepressants ultimately raise serotonin levels, so does MDMA.
MDMA
MDMA is a sneaky bastard. Insidiously it takes control of the infrastructure and turns the whole system upside down. How does it do that? First, MDMA enters the neuron via the serotonin transporters (SERT). Once inside the neuron, it inhibits the vesicular transporters (VMAT2) which means that serotonin is not neatly packed within the vesicles anymore, but now accumulates within the cytosol. Then, MDMA reverses the direction of the SERT, meaning instead of transporting serotonin into the neuron, they now release it into the cleft and deny its reuptake.13 The result is a dramatic increase of serotonin levels in the synaptic cleft which makes the receptors on the postsynaptic membrane go haywire for a few hours.
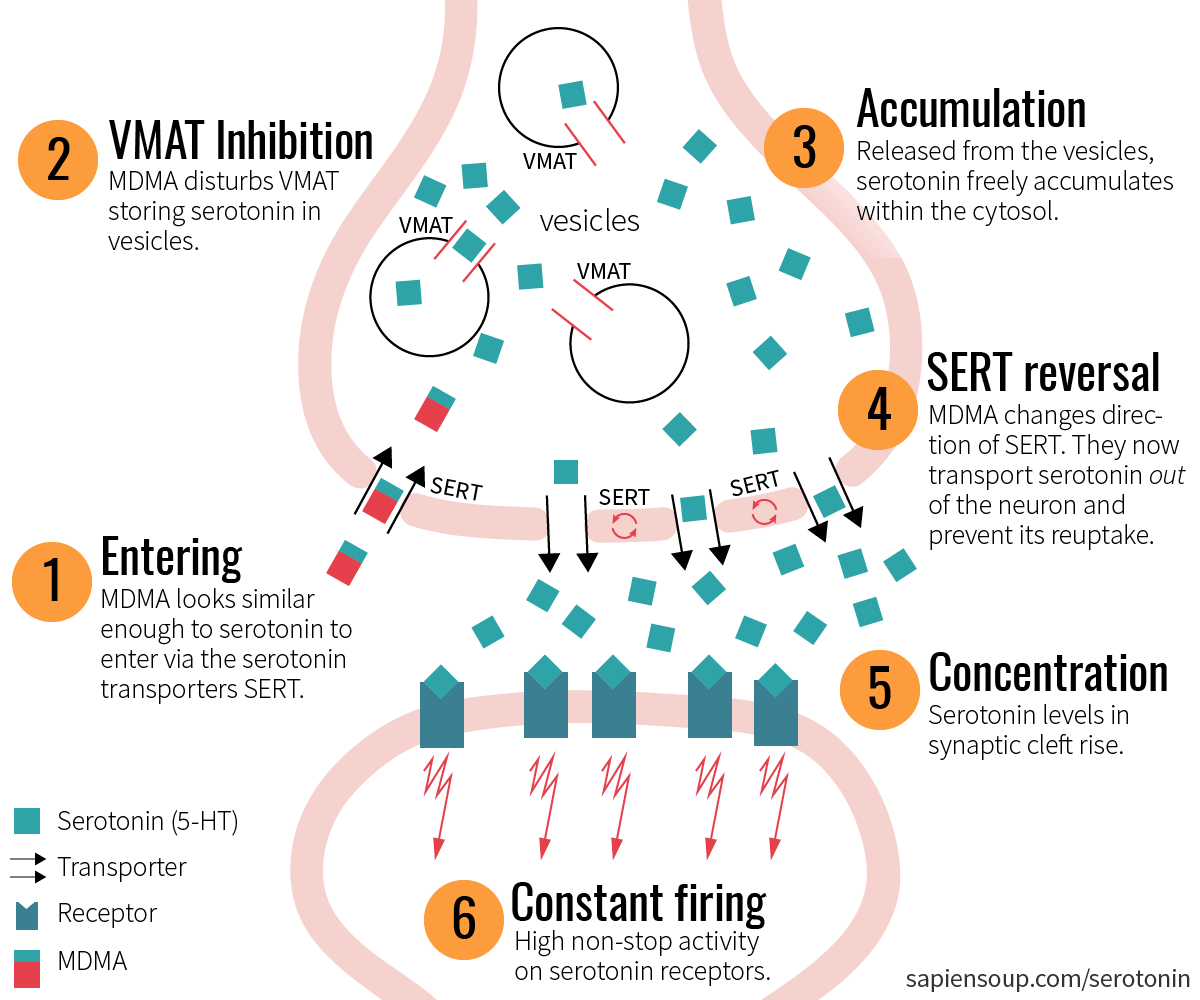
Moreover, MDMA increases dopamine and norepinephrine (i.e. noradrenaline) levels, which gives it its ecstatic properties. This temporary overstimulation of the serotonergic system leaves the neurons depleted of serotonin and needing to recover after the drug use.
Why it doesn’t cause addiction
What if MDMA is taken daily to keep up colossal serotonin levels? The short answer is: doesn’t work; the effect of MDMA is capped by the available serotonin. If the brain is depleted from serotonin, MDMA has no material to work with and therefore the effects would be rather disappointing for the user. The brain needs time to replenish its supply before the drug could achieve the desired effects once more. Many users feel irritable and depressed after using MDMA.14 But when using again is not an option, there isn’t much of an addiction loop they could tap into. With this built-in mandatory refractory period, the physical addiction potential of psychedelic drugs is limited.
With a built-in, mandatory refractory period, the physical addiction potential of psychedelic drugs is quite limited.
Remember how the brain usually doesn’t release large quantities of serotonin at once? Other neurotransmitter systems in the brain are more suitable for this task: dopamine for example. The dopaminergic system does react well to repeated stimulation and is therefore frequently involved in addiction. Drugs which target the dopaminergic system are cocaine, amphetamine, methamphetamine, but also Adderall and Ritalin.15
LSD, psilocybin and other hallucinogens
Unlike MDMA, hallucinogens don’t flood the brain with serotonin. They target a specific subtype of serotonin receptor—the 5-HT2A receptor—to which they bind directly, thereby activating it. The 5-HT2A receptor is known to play a key role in regulating mood, anxiety, schizophrenia and consciousness.
There is so much to say about how hallucinogens affect the brain. The initial hypothesis—that hallucinogens increase the activity in certain areas of the brain—was recently abandoned. In fact, hallucinogens temporarily shut down some major connecting hubs.16
Why does this stir a researcher’s blood? Because if you want to know what a certain area in the brain does, it helps to observe what goes missing if you shut it off. Turned out, shutting off those connector hubs led to the interruption of the brain’s default mode network (DMN).17 You can think of the DMN as something like a screensaver which randomly shuffles through images of your past, your future, your to-do list, the super size menu that you shouldn’t have eaten, the sad face of a person you hurt and so on. Interrupting the DMN has very interesting consequences which we will—at length—cover in a future post. It’s a phenomenally interesting topic that deserves its own post (and requires a couple of thousands more words to explain).
Also, when breaking up the regular communication pathways, the brain starts to communicate in brand new ways. This visualization shows brain regions communicating which one another in (a) a normal state or (b) after administering psilocybin. On the left you can see that the color-coded regions communicate mostly amongst themselves, i.e. the dots of the purple region talk to other dots within the purple region. But under the influence of an hallucinogenic drug the purple dots start talking to all kinds of other brain regions.
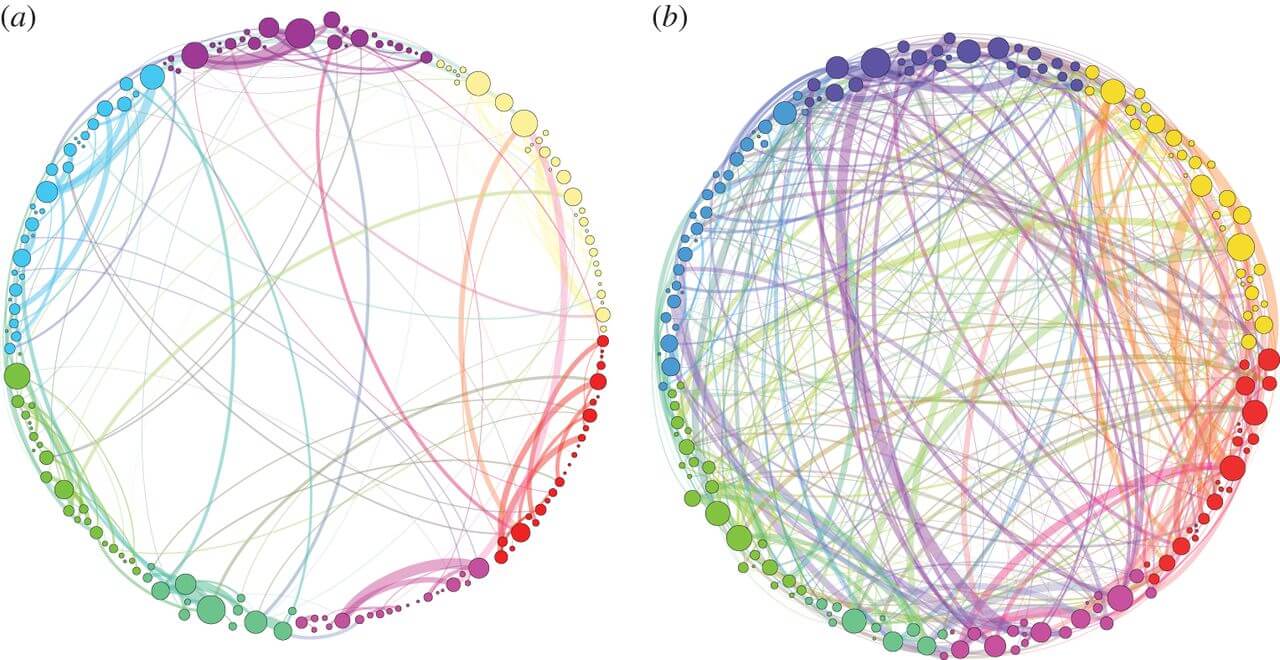 Communication pathways in the brain after (a) placebo and (b) psilocybin18
Communication pathways in the brain after (a) placebo and (b) psilocybin18
These novel communication pathways might be able to explain the creativity-enhancing and problem-solving qualities that are often attributed to hallucinogenic drugs.
The problem with criminalization
What’s the essential nature of science? (1) You find an interesting thing, (2) you test and observe how the thing behaves under different conditions and (3) you come up with a hypothesis.

Psychedelic drugs and the serotonergic system are deeply intertwined. Not only was LSD involved in the initial discovery of the serotonergic system which later revolutionized psychiatry.2 Today, psychedelic research could yet again revolutionize our understanding of the human brain. Manipulating 5-HT2A receptors has astounding effects on brain circuitries that are involved in the sense of self and consciousness. You can think of the 5-HT2A receptor as the little kid’s basket and hallucinogens as a potent tool to test it. Psychedelic drugs might be nothing less than our key to deciphering consciousness.
With brand-new imaging technology, we could now watch the brain as it loses its sense of time and space. We could observe which regions fall out of sync when it dissolves its sense of self. We could literally watch the brain as it changes its state of consciousness. The problem is, we’re not allowed to. The current drug legislation makes psychedelic research so difficult and so expensive, that only very few research teams manage to get approval and funding for their studies.
Criminalization of psychedelic drugs stands between science and the exploration of consciousness.
A mighty and mysterious system
Having discovered the serotonergic system less than 70 years ago, there is much that remains unknown about this mighty and mysterious network of neurons. We know that it is crucial for a lot of processes, but the ins and outs are not well understood even today. It will be a long time before we’ll figure out the exact mechanisms of this versatile system.2
In the meantime, every new study on psychedelics reveals fascinating new insights about consciousness, the brain’s default mode network and mental disorders. Over the next posts in our Psychedelic Drugs series we’ll cover the outcome of those recent studies.
Medical Benefits of Psychedelic Drugs
Psychedelic Drugs and the Serotonergic System (You’ve just read it)
The Psychedelic Experience
Your Brain on Psychedelic Drugs
Psychedelics and Mental Health
Microdosing LSD: Smart Drug or Placebo?
MDMA-assisted Therapy
Wow, that was a lot of information to take in. But you did it! Now you know more about the serotonergic system than any of your friends (except if your friends are neuroscientists). Since you seem to be really interested in the topic you might want to get notified when we publish our next story.
If you are located in the Vienna area, we invite you to join the Psychedelic Society Vienna meetup, where we’ll discuss the latest research and developments in the field.
References
-
David E. Nichols. LSD Neuroscience. 2013. Youtube. Retrieved 2017-05-25. ↩ ↩2
-
Nichols DE. Serotonin, and the Past and Future of LSD. MAPS Bulletin Spring 2013. Retrieved 2017-05-26. ↩ ↩2 ↩3 ↩4 ↩5
-
Mann JJ. Role of the serotonergic system in the pathogenesis of major depression and suicidal behavior. Neuropsychopharmacology. 1999 Aug;21(2 Suppl):99S-105S. Review. PubMed PMID: 10432495 ↩
-
Sodhi MS, Sanders-Bush E. Serotonin and brain development. Int. Rev. Neurobiol. 2004;59:111–74. PubMed PMID: 15006487 ↩
-
Illustration from Wikimedia Commons. Modified from Paradiso MA, Bear MF, Connors BW. Neuroscience: exploring the brain. 2007. Hagerstwon, MD: Lippincott Williams & Wilkins ↩
-
Bear M, Connors BW, Paradiso MA. Neuroscience. Exploring the Brain. 4th edition. Wolters Kluwer. ↩ ↩2 ↩3
-
Zafeiriou D., Ververi A, Vargiami E. The Serotonergic System: Its Role in Pathogenesis and Early Developmental Treatment of Autism. Current Neuropharmacology. 2009;7(2):150-157. PMC2730007 ↩
-
Serotonin transporter. Wikipedia. Retrieved on 2017-05-22. ↩
-
Reneman L, Endert E, de Bruin K. The acute and chronic effects of MDMA (“ecstasy”) on cortical 5-HT2A receptors in rat and human brain. Neuropsychopharmacology. 2002 Mar;26(3):387-96. PubMed PMID: 11850153. ↩
-
5-HT receptor. Wikipedia. Retrieved on 2017-05-22. ↩
-
Refrigerator mother theory. Wikipedia. Retrieved on 2017-05-26. ↩
-
Whitaker-Azmitia PM. The discovery of serotonin and its role in neuroscience. Neuropsychopharmacology. 1999 Aug;21(2 Suppl):2S-8S. PubMed PMID: 10432482 ↩
-
Eiden LE, Weihe E. VMAT2: a dynamic regulator of brain monoaminergic neuronal function interacting with drugs of abuse. Annals of the New York Academy of Sciences. 2011;1216:86-98. PMCID: PMC4183197 ↩
-
MDMA Basics. Erowid. Retrieved on 2017-05-22. ↩
-
David Nutt. Psychedelic Research, From Brain Imaging to Policy Reform. 2017. Youtube. Retrieved 2017-05-29. ↩
-
Carhart-Harris RL, Muthukumaraswamy S, Roseman L, et al. Neural correlates of the LSD experience revealed by multimodal neuroimaging. Proc Natl Acad Sci U S A. 2016 Apr 26;113(17):4853-8. PubMed PMID: 27071089. ↩
-
Petri G, Expert P, Turkheimer F, et al. Homological scaffolds of brain functional networks. J R Soc Interface. 2014 Dec 6;11(101):20140873. PubMed PMID: 25401177. ↩